Glossary
Everybody knows and uses batteries, but they are not quite trivial. From the chemical processes that take place, to the components contained, to the various uses, everything has its technical terms and special features. But you don't have to make it unnecessarily complicated. We have tried to break down the most important terms here.
Absorbent Glass Mat
Battery class in which the electrolyte is bound in a glass fibre mat separator.
Because the electrolyte is fixed, an AGM battery cannot leak. It is also vibration-resistant because the separators swell after they have absorbed the electrolyte, thus protecting the grid plates from vibration and material loss. Extremely vibration-resistant heavy-duty batteries are often based on advanced AGM technology.
In addition, because the glass fiber mats lie close to the electrodes, less active electrode material is lost during each discharge-charge cycle than in conventional batteries.
AGM batteries are used in cyclical and stationary applications such as telecommunications, wheelchairs, alarm systems as well as in classic starter applications and start-stop applications.
If your car with start-stop technologies had an AGM installed, you should definitely replace it with an AGM start-stop battery. An upgrade from EFB technology to AGM is always possible. Both start-stop batteries must be registered in the car's management system after installation.
Acid
The electrolyte used in lead-acid batteries. In GEL batteries it is a sulphuric acid (H2SO4) thickened with silica.
In AGM batteries, it is a sulphuric acid that is absorbed in a glass mat. In flooded lead-acid batteries, the electrolyte is in liquid form and is a sulphuric acid diluted with battery water. Their concentration, and therefore their density, changes with the state of charge, so by way of an acid siphon it is possible to determine whether a flooded battery is charged.
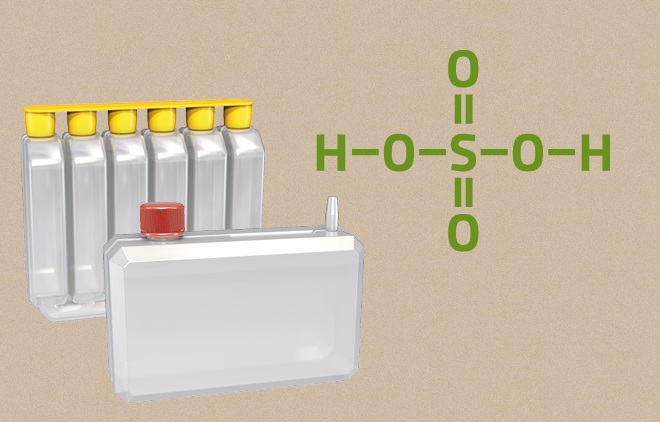
Acid Pack
A quantity of battery acid exactly adjusted to the filling quantity of a dry-charged battery.
Since the EU Regulation VO (EU) 2019/1148 came into force on 1.2.2021, this acid pack may no longer be issued to the user.
You can still obtain our dry-charged motorcycle batteries from specialist dealers. They will be properly filled there for you with the acid pack and charged before installation.

Acid Stratification
An undesirable process in batteries where the heavy sulphuric acid settles in the lower part of the battery and the water on top.
This can be caused by incorrect charging and can be avoided by correct charging.
If the acid is stratified, chemical processes only take place in the lower part of the battery, the performance decreases and the battery fails within a very short time. It can aggravate plate corrosion, sulfation and the formation of battery sludge.
Active Material
The chemical pastes which, in combination with the battery acid, produce electric current.
They adhere to a current collector made of lead or a lead alloy (often in the form of a grid) and together with it form the positive (+) and negative (-) plates of a battery. On the positive plate the active material is lead dioxide (PbO2), on the negative plate lead.
The more active material there is, the greater the amount of current a battery can supply. This is crucial for the cold start capability of batteries. For this reason, current collectors are built in a grid shape or, as with the armor plate, consist of numerous lead tubes with the largest possible surface area.
Advanced Flooded Battery
Outdated term for a category of start-stop batteries. New: EFB
Alloy
A mixture of a metal with other metals or other substances to change its properties.
The current conductors in batteries are made of pure lead or lead alloys. Pure lead is very soft and can only be used in wound or tightly packed batteries.
In the past, the chemical element antimony was often used for hardening, but it is not suitable for sealed batteries. Using a calcium alloy, which is common today, also significantly reduces water loss during the chemical processes in a battery. This in turn makes it possible to manufacture sealed, leak-proof and absolutely maintenance-free batteries.
Ammeter
A measuring device to determine the current intensity.
Amp-Hour
The unit of measurement for the electrical storage capacity of a battery.
Ampere
The unit of measurement for the amperage of a battery.
Average Drain
The average amount of current drawn from a battery during discharge.
It is usually approximated by calculating the current at a discharge depth of 50 %.
Backfire Protection
A safety feature that prevents a spark from entering the battery from the outside.
Sparks can be caused by static charging or when disconnecting the battery from the charger, or even by open fire.
Backup Battery
A battery that ensures the car's electrical functions in case the main starter battery is causing problems.
It also serves to relieve the main battery when the car has many electrical consumers that require power before the engine is started.

Balancing
The process that ensures that all cells in a battery are and will be evenly charged.
Batteries consist of several cells. Due to different aging or tolerances in manufacturing, they may discharge at different rates or charge at different rates. Lithium batteries have a battery management system. In this case, an electronic circuit, the balancer or equalizer, ensures that the charge is nonetheless evenly distributed. This allows the total capacity of the battery to be used in the best possible way and, at the same time, protects the individual cells against overcharging.

Battery
An electrical energy source consisting of one or more primary (non-rechargeable) or secondary (rechargeable) cells.
Strictly speaking, a single cell is not a battery and a rechargeable battery is called an accumulator. In common usage, the term battery is nevertheless also used for monocells and rechargeable accumulators such as car batteries. When we speak of battery here, we almost invariably mean rechargeable accumulators.
Battery Care
All measures we recommend to counteract the ageing process of a battery.
This includes, for example, correct commissioning, regular checking of the state of charge, charging at sensible intervals, cleaning the poles and the battery.
Every battery benefits from appropriate battery care.
Some batteries require additional maintenance, such as checking the acid level or refilling with distilled water if necessary.
Battery Case
The outer shell of a battery.
Classic wet batteries are often in a white or transparent case so you can easily check the fluid level.
Some cases have a bottom bar to keep them securely in place. There are different standards for the box sizes. The exact dimensions of our batteries are noted with each product.
Battery Categories
Batteries categorised according to common characteristics.
In principle, all batteries are galvanic cells in which voltage is created in the electrolyte by the chemical reaction at the two poles. Batteries have very different characteristics in the design of the housing, the separators, the alloy of the grids and active material, the composition of the electrolyte, etc. For example, AGM batteries have a high cold-start performance.
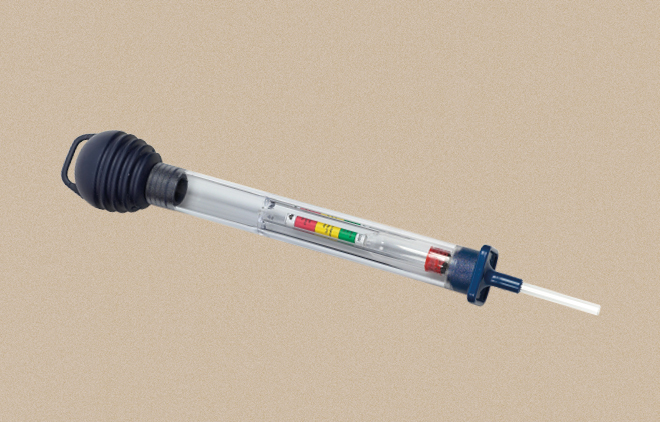
Battery Hydrometer
A measuring instrument for determining the density of liquids.
For batteries, their state of charge can be determined by the density of the electrolyte. For this purpose, electronic hydrometers or mechanical float devices are used.
Boost Charging
Charging with high charging currents.
Calcium Battery
A battery in which both grids are made of a lead-calcium alloy.
The calcium alloy has largely replaced the lead-antimony alloy that was previously used. The use of calcium reduces the amount of gas in the battery. Less liquid is lost, making closed, maintenance-free batteries possible. Calcium batteries are mostly used as starter batteries because they have a good cold start performance. They are less suitable for cyclical use, i.e. frequent charging and complete discharge, as when used as a drive or supply battery.
Capacity
The amount of current that a fully charged battery can deliver.
It is measured in ampere-hours (Ah) and indicates how many hours (h) a battery can supply current at a certain level (Ampre/A). Therefore, the greater the consumption and the longer the vehicle or device is to be operated before it is recharged, the higher the Ah capacity of the battery must be.
This is especially important for traction batteries or supply batteries.
Carbon Additive
An addition of carbon to the lead alloy of the electrodes.
This increases the starting performance and cycle stability of a wet battery, both of which are used, for example, in the EFB battery.
Cell
A set of positive (+) plate, negative (-) plate, electrolyte, separator and housing.
In lead-acid batteries, several cells of 2.12 V each are combined to obtain a nominal voltage of 6, 12 or more volts. Secondary cells can be recharged, primary cells cannot.

Cell Short-Circuit
A short circuit between the plates of one cell.
A plate closure can be detected by measuring the voltage of a battery. If the voltage is lower than the nominal voltage exactly by the value of one cell, this is most likely due to a plate short circuit. The battery is irreparably destroyed.
There are several possible causes for direct contact between negative and positive plates, i.e. a short circuit: excessive sulphation causes the plates to grow too much and damage the separators; mechanical stress causes the grids to bend; corrosion causes the grids to grow excessively; battery sludge forms an undesirable bridge between plates of different polarity.

Charge Regulator
An electronic component in vehicles that controls the voltage generated by the alternator and protects the battery from overload or reverse current.
Charger
A device for recharging accumulators.

Charging
The reversal of the chemical process that occurs when generating the current or when discharging. Rechargeable batteries are strictly speaking called accumulators or secondary batteries.

Charging Voltage
The voltage used to overcome the internal resistance of a battery and charge the battery.
If the voltage is too low, charging will take a long time, too high a charging voltage can cause permanent damage to the battery through grid corrosion. Intelligent chargers prevent this.
Circuit
The path that the electrons take when current flows, i.e. the positive and negative terminals are connected to each other via electrical consumers or a charger.
When the battery is discharged, electrons flow from the negative plate, through the negative terminal, via the current consumer, to the positive terminal, to the positive plate and via the battery acid to the negative terminal. When charging, the reverse is true.
Closed Battery
A battery that is closed but has no valve.
The partly used term "open" battery is misleading, because also a so called "open" battery is closed by plugs or similar. However, it can be opened for maintenance (low-maintenance battery). Maintenance-free closed batteries are closed at the factory and cannot be opened. They usually have a central degassing system or special lids through which only very little gas escapes under normal circumstances. In contrast, the sealed battery (VRLA) is valve-controlled.
Cold Cranking Amps (CCA)
The value in amperes that a new, fully charged battery can deliver at a temperature of -18 °C for 30 seconds before the voltage becomes too low.
The cold start performance indicates whether a battery can provide the necessary power even in winter, when the electrochemical processes are slower. This is important for starter batteries in vehicles with many consumers or when selecting supply batteries that are also to be used at low temperatures.
Constant Current Battery Charger
A charger that charges a battery with a current that remains constant over the charging period.

Constant Current Discharger
The discharge of a battery by a constant current.
Constant-Voltage Battery-Charger
A charger that regulates the charging voltage based on the state of charge.
Continous Test
A process in which a battery is constantly drawn current up to a certain point.
So the existing capacity of the battery is determined.
Corrosion
A chemical reaction in which metal is decomposed, in batteries an undesirable aging process.
Corroded battery terminals, battery clips and terminal adapters conduct the current only very poorly or not at all. To avoid battery failure, it makes sense to check the terminals regularly and remove any corrosion that may occur.
Also the grids of the positive plate in a battery can corrode and even break. This usually happens when the charging voltage is too high, when the temperature is too high, when the acid density is too high, or when there is acid stratification.

Current
The amount of electricity a battery can supply, measured in amperes.
A suitable battery provides enough electricity to supply all consumers of the vehicle or appliance. How long it can do this depends on its capacity.
Current Collector
A component of an electrode to which the paste-like active material for the chemical reaction adheres.
The current collector gives stability to the active material and conducts the current to the outside. In lead-acid batteries it consists of pure lead or lead alloys.
The current collector forms a plate together with the active material.
If one speaks of grid plate, tube plate etc., strictly speaking it is about the shape of the current collector.
Cutoff Voltage
A value specified by the manufacturer up to which a rechargeable battery can be discharged without affecting its service life.
If the battery were to be discharged even further (deep discharge), there is a risk of damaging it. Flooded lead-acid accumulators are very sensitive to deep discharge.
Cycle
The cycle of a rechargeable battery, from the charged battery to the complete discharge to the complete recharge.
Traction batteries, e.g. of lifting platforms, are normally completely discharged and only recharged afterwards. How well they withstand this determines their cycle stability and thus their service life.
Cycle Life
The number of cycles a battery can go through before its capacity falls below a certain nominal value.
When the battery can only be charged to 60 % of its initial capacity, it has reached the end of its cycle life.
Cycle stability is important for batteries used to power vehicles or appliances, for stationary supply and for the temporary storage of solar or wind energy.
Cyclic Loads
Consumers, which usually draw a constant amount of current until the battery is empty and is recharged.
For example an electric wheelchair or a refrigerator in a motor home or on a boat. The ignition in a car, on the other hand, is a consumer that needs a lot of power for a short time.
Cycling
The repeated passing through of a cycle, for batteries in the cycle of discharging and charging.
Some batteries are designed to cycle, such as utility or traction batteries.
Cylindrical Battery
A battery consisting of cylindrical cells with tightly wound lead plates.
In contrast, the usual lead-acid batteries consist of cells with plate sets of flat lead grids.
Through tight winding you get a large surface in a very small space, so a good starting performance. Round cell batteries use AGM technology, i.e. the electrolyte is bound in glass fibre fleece. This gives these batteries good cyclical behaviour. They are suitable as supply batteries in buses and motor homes, on boats and in solar and wind power systems in the semi-professional sector.

Damage due to Long Down-Times
An undesirable aging process that occurs when the battery is not charged for a long period of time.
Sulfation is a typical damage caused if a battery is kept sitting too long without proper care and charging. The capacity of the battery drops significantly, or it fails completely.
Deep Discharge
The state in which a battery is 80 percent or more discharged.
We also speak of a discharge depth of 20 %.
Lithium batteries withstand this particularly well.
Deep-Cycle Battery
A battery that is designed to withstand repeated discharges to a depth of 20 % or more and continue to provide its rated capacity even after hundreds of cycles.
deep cycle batteries are often used in boat, camper and industrial applications.

Degassing
The positioning of the vent openings in a battery according to the vehicle manufacturer's specifications.

Depth of Discharge
The degree to which a battery is discharged.
Usually the manufacturer specifies a maximum depth of discharge to which the battery can be discharged without impairing its life.
Direct Current
An electric current that runs with constant strength in the same direction.
A battery supplies direct current. It is a direct voltage source, i.e. it does not change its polarity.
In contrast, a voltage source with alternating polarity provides alternating current.
Discharge Rate
A measurement number that determines how long it takes for a battery to be completely discharged or charged.
A discharge rate of 1C stands for a 1-hour discharge, 2C for 1/2-hour discharge, 10C for 6-minute discharge, etc. The discharge rate is independent of the capacity of a battery.
Analogue, 1C also stands for 1 hour charge, 2C for 1/2 hour charge etc. Here, this is the time required to fully charge the battery.
Discharging
The process of drawing power from a battery to supply electrical energy to connected loads.
In addition to deliberately switched-on electrical loads such as heating or lighting, components or devices such as alarm systems or computer memory, so-called silent loads, also draw power from the battery while a vehicle is not in use.
A battery that is no longer fully charged is called discharged; if it is 50% discharged, it is called deep discharged.
Distilled Water
Distilled water to refill batteries that lose fluid.
Classic lead-acid batteries, also known as wet batteries, lose fluid during operation. The liquid level must therefore be checked regularly and topped up if necessary. Distilled water is required for this purpose so as not to change the composition of the acid. Unsuitable water would destroy the electrodes within a very short time, rendering the battery unusable. Maintenance-free batteries cannot be refilled with battery water.
Double Lid
A lid shape in which the water vapour produced condenses in the labyrinth of the lid and is returned to the battery.

Dry-Charged
Designation for a battery that is dry-charged on delivery but does not yet contain acid.
This battery must be filled by a specialist dealer before use. It may only be charged after a waiting period (at least 30 minutes) and only closed afterwards. Some AGM motorcycle batteries are dry-charged when delivered. After initial filling and closing, they are maintenance-free and cannot be opened.

Electrolysis
The chemical process that generates electric current in a battery.
Electrolyte
A necessary component of batteries, in conventional lead-acid batteries usually a diluted sulphuric acid, which is electrically conductive due to the ions it contains.
Electrolytes used for power generation are often liquid, but their properties can be changed by various additives. In gel batteries, for example, the sulphuric acid is bound in a gel-like manner by adding silicic acid. This battery class loses almost no liquid and is therefore maintenance-free or absolutely maintenance-free. In flooded batteries, on the other hand, the electrolyte level must be checked regularly and refilled with battery water if necessary.
End of Charging Voltage
The maximum electrical voltage specified by the manufacturer to which a single cell may be exposed during charging.
If it is exceeded, the cell can be irreparably damaged. When charging via a charger, the final charge voltage is applied to both poles.
Enhanced Flooded Battery
An improved flooded lead-acid battery designed to withstand significantly more cycles.
To achieve this, carbon additives are added to the lead and the electrodes are surrounded by a special separator pocket.
EFB batteries are intended for use in vehicles with start-stop technology, electrically when the car stands and is restarted when the vehicle is moving off. They also support vehicles with energy recovery, where the battery is charged, for example during braking. Thanks to their high cold-start performance, however, they are also suitable as starter batteries in cars with many consumers.
They also withstand high temperatures well and are therefore suitable in hot climates or for installation directly in the engine compartment - even in vehicles with conventional drive systems.
In addition to EFB batteries, AGM batteries are also installed in vehicles with start-stop function. If an AGM battery is installed at the factory, it may only be replaced by an AGM battery. An upgrade from EFB technology to AGM is always possible. Both start-stop batteries must be registered in the vehicle management system after installation.

Expanded Metall Grid
A variant of grids produced by stretching the metal used.
Fill Level
Marking on the outside of the white or transparent casing of a wet battery indicating the minimum (MIN) and maximum (MAX) filling level of the battery.
During commissioning or initial filling, the battery must be filled with battery acid up to the upper mark (MAX). During subsequent maintenance, the battery should be refilled with distilled water up to the MAX mark.

Flat Plate
A plate consisting of a grid frame and active material.
The grid shape of the frame means that it can hold a particularly large amount of active material. This increases the capacity with a comparatively small volume achieved.
more info → plate

Float Charge
The low, constant charging of a fully charged battery to compensate for self-discharge.
This ensures that the battery is always fully charged. This is especially important for batteries that are in constant standby, e.g. to be able to take over in case of failure of other power sources.
Flooded Battery
A category of lead-acid batteries that can be operated with diluted sulphuric acid, which is in liquid form, i.e. not immobilized as GEL or held glass fibre mats.
They are available in two versions: Low-maintenance and maintenance-free. In the case of the low-maintenance battery, it is necessary to regularly (every 2 to 3 months) check the state of charge and the fluid level and, if necessary, refill with distilled water. It can be identified by the removable plugs and the venting hose. It can only be installed upright, otherwise it will leak. Maintenance-free flooded batteries, on the other hand, cannot be opened or refilled. Compared to the low-maintenance flooded battery, they are more leak-proof.
Until about 1995, vehicles were equipped with alternators designed for flooded batteries. These vehicles cannot be equipped with a different battery class. If in doubt, replace an existing flooded battery with a flooded battery again.
Fuse
A circuit breaker or other interrupting device in a circuit.
If the current exceeds a certain value, it interrupts the circuit to protect it from short circuits or current surges.
Gas Recombinant Technology
A technology in which most of the gas produced in batteries is liquefied again and fed back into the chemical cycle.
Small amounts of excess gas are discharged via a central degassing system or via a valve to avoid overpressure.
Gas recombination is the key to maintenance-free batteries.
Gassing
Designation for the gas development in a cell while electricity is generated by chemical processes.Energy
When the batteries are sealed, only a small amount of gas is produced, which escapes through safety valves in case of overpressure. In wet batteries with higher gas development, it escapes via one or more vents and is conducted outside the engine compartment via a vent hose. On vehicles from different manufacturers, the vent openings and possibly hose outlets are also located at different points in the battery, e.g. on the right, left or both sides.
In flooded batteries, gas generation during charging is wanted, as the rising gases mix the acid and counteract acid stratification.
Gel Battery
A class of batteries in which the electrolyte is thickened to a gel-like consistency by the addition of silica.
Gel batteries are sealed and have a safety valve. This makes them leak-proof, absolutely maintenance-free and more vibrant resistant than flooded batteries.
They have a very low self-discharge rate and can withstand deep discharge.
They are used as supply and traction batteries and as starter batteries in vehicles with start-stop technology or motorcycles with many short trips.

Glass Mat
A material from which separators in batteries are made. In AGM batteries, the fibreglass mat holds the electrolyte.
Grid
A grid-shaped current collector made of lead or a lead alloy.
Together with the active material adhering to it, it forms a plate in a battery. Apart from a grid, a current collector can have various other shapes.
The grid gives the active material support and also conducts the current to the outside. All grids with positive material form the positive pole when connected together, all grids with negative material form the negative pole.
Depending on the alloy from which this grid is made and how it is mechanically produced, e.g. rolled or twisted from wire, different battery properties result. These in turn influence, for example, whether a battery is particularly good at storing current, can be charged quickly or can deliver a lot of current very quickly (high cold-start performance).

Ground
A large conductive body, such as the metal frame of a vehicle.
For one thing, it provides safety when it is connected to the ground via a ground cable. On the other hand, the ground is the reference point for calculating the voltage in an electric circuit.
Today in vehicles almost exclusively the negative (-) connection of the battery is used as ground.
Ground Cable
The cable connecting the battery to ground, e.g. the metal frame of a vehicle.
Normally the grounding cable is attached to the negative (-) terminal. Some older English or US vehicles use the positive (+) terminal as the grounding terminal.
Grounding is a safety measure against overvoltage.
HVT
A battery category designed to meet the unique needs of Harley-Davidson motorcycles. The HVT is extremely robust to ensure reliable start-up under continuous load caused by the severe vibrations of Harleys and other large-capacity 2-cylinder engines. The plate packs are highly compressed by special separator pockets. In addition, they are perfectly fitted in the robust and impact-resistant housing.

Hybrid Battery
A lead-acid battery in which one grid is made of a lead-calcium alloy and the other of a lead-antimony alloy.
Immobilized Electrolyte
Term for a non-liquid electrolyte.
Here, the liquid acid has been gel-bonded by adding an additive such as silica or rendered immobile by glass fiber mat separators.
Internal Resistance
A measurement to sum up all internal resistance in a battery.
Each voltage source has an internal resistance. In detail, the internal resistance is composed of the polarisation resistance of the electrochemical reaction (the energy required for the chemical process), the flow resistance of the ions and the ohmic resistance of the electrodes. The electrical equipment therefore does not receive the entire terminal voltage, as a part of it is lost in the internal resistance of the battery. If the current flow increases, the voltage drop at the internal resistance grows and the voltage decreases.
Ion
An electrically charged atom or molecule.
Lead Sediments
An unwanted ageing process whereby tiny pieces of active material come off with each charge cycle and settle at the bottom of a battery.
To prevent this conductive battery sludge from touching the plates, they normally do not reach the bottom of the battery. If the battery sludge reaches the plate, a short circuit can occur that renders the cell unusable. This is also referred to as plate short circuit.
Lead Sulfate
Produced in lead-acid batteries when generating electricity from sulphuric acid and lead.
When charging the battery, the lead sulphate is removed again. If too much lead sulphate is produced, the battery sulphates.
Lead-Acid Battery
A rechargeable battery that, by means of electrolysis, generates electric current by combining lead and lead oxide as the active material and an acid as the electrolyte.
Depending on the materials used and how they are combined, a distinction is made between standard flooded batteries, AGM batteries, GEL batteries and many other types.
Lithium Iron Phosphate Battery
A lithium-ion battery in which the active material of the positive (+) terminal consists of lithium iron phosphate.
This version of the lithium-ion battery has become established in the supply and motorcycle sectors because, unlike some Li-ion variants, it is considered to be extremely safe. In the event of overcharging, no metallic lithium is deposited in LiFePO4 and no oxygen is released, which significantly reduces the fire hazard.
LiFePo4 batteries, like all lithium-ion batteries, are very small and light and have a long service life. They can be installed in any position, since they do not contain liquid electrolyte. They have a low self-discharge, tolerate the highest charging currents and can be charged very quickly. Special lithium chargers or the simplest chargers with automatic cut-off are suitable for charging. Modern pulse chargers are not suitable for lithium-ion batteries.
Lithium-Ion Battery
The generic term for batteries in which both terminals and the electrolyte contain lithium ions.
Depending on the material used for the terminals, the electrolyte and the separators, LIBs have very different properties. What they all have in common is that their capacity and performance is very high compared to other batteries. They are mainly used when size and weight are important, e.g. in racing and motor sports training sessions or when particularly high power is required, e.g. when a large number of loads need to be supplied.

Load
Components or devices that draw their energy from the power source provided.
Low-Maintenance
Term for a conventional flooded battery that requires regular maintenance, at least before the winter break.
Here, the fluid level is checked and, if necessary, refilled with distilled water.

M.A.S.S.
A safety feature of intAct batteries that prevents electrolyte leakage, even during prolonged upside down position (rollover test).
The housing covers specially designed for the safety system M.A.S.S. contain new types of labyrinth seals and an integrated backfire protection.
Maintenance
Regular checks of the liquid level and refilling with distilled water if necessary.
Unlike maintenance-free batteries, classic lead-acid batteries require regular maintenance, at least before winter. This involves checking the fluid level and refilling with battery water if necessary. It is also good for all batteries to check them regularly for external damage and corrosion and to check their state of charge.
Maintenance-Free
A battery where under normal operating conditions no battery water needs to be refilled and which cannot be opened.
Not to be confused with a sealed, absolutely maintenance-free battery.

Memory Effect
The loss of capacity that occurs when batteries are recharged after a short discharge.
With lead acid batteries there is no memory effect. Especially starter batteries are only slightly discharged during normal operation before being recharged by the alternator during the journey.
Multimeter
An electronic measuring device that can determine several measured variables, including voltage, current and resistance.

Negative (-)
One of the two poles of a battery.
The other one is the positive pole. The cap on the negative pole, the negative terminal and the negative cable are usually black. In order for current to flow, both the negative and the positive terminal must be connected to a load.
Negative (-) Plate
The plate on which oxidation occurs when the battery is discharged as soon as plus and minus are connected to a load.
The electrons produced during oxidation are taken up by the grid of the negative plate, transferred to the negative pole via the pole bridge and then to the current consumer.
This process is reversed when charging the battery.
In lead-acid batteries, box or grid plates are usually used for this purpose, in whose meshes the lead is deposited.
In a rechargeable battery, the negative (-) plate is called the cathode (strictly speaking, the designations anode and cathode depend on the chemical processes and thus the direction of current flow during charging and discharging, but this could lead to misunderstandings, so the designation is determined by the way the current flows during discharging).

Nominal Voltage
The voltage that a battery provides nominally, i.e. according to its designation.
Batteries must be selected so that the nominal voltage matches the voltage required by a vehicle or device in normal operation. For motorcycles this is normally 6 or 12 volts.
A battery with a nominal voltage of 12 V actually delivers a voltage of 12.72 V, consisting of 6 cells connected in series at 2.12 V each. The open-circuit voltage is measured when the battery has been disconnected from any charge for at least 4 hours and no current has been drawn for at least 1 hour.
Non-Spillable
A battery that is firmly sealed and equipped with valves.
This means that no liquid can be refilled, which is why these batteries are referred to as sealed maintenance-free. With AGM and HVT batteries the electrolyte is held in a non-woven mat and they are therefore leak-proof. Just like GEL batteries, where the electrolyte is in the form of a gel. Both can be installed at an angle. In some areas, leak-proof batteries are mandatory, e.g. on boats, to prevent battery fluid from escaping in the event of capsize.
Open Circuit Voltage
see → Open circuit voltage
Open-Circuit Voltage
The voltage of a battery at rest, measured at least 4 hours after the last charge and after no current has been drawn for at least 1 hour.
The resting voltage provides information about the condition of a battery and notes on care.
To avoid damaging your battery, it should always be charged at least 75 %. For a battery with a nominal voltage of 12 V, 100 % charge corresponds to 12.69 - 12.75 V, 75 % corresponds to 12.51 - 12.57 V. If the battery is to be constantly fully charged in order to be available for certain tasks in case of emergency, it should be kept permanently at 0.1 V per cell above the nominal voltage. For a 12 V battery, this corresponds to a total voltage of 13.2 V.
If the open-circuit voltage of your battery is too low by the voltage of one cell, this indicates the plate short, i.e. the defect of a cell. This is probably a warranty case. In this case contact your dealer.
Overcharging
An improper charge in which the battery continues to charge after reaching the end-of-charge voltage.
The heat generated by overcharging the battery can lead to deformation and even explosion of the battery box.

Parallel Circuit
An arrangement of cells in which all negative (-) poles are connected to each other and all positive (+) poles to each other.
In such a circuit the capacity of the individual cells adds up to the capacity of the whole battery. The voltage is determined by the weakest member of the series, or in this case the weakest cell.
The opposite is the series connection or series circuit.
Placement of Terminals
The arrangement of the positive and negative terminals for batteries is called a circuit.
It has to match the vehicle type.

Plate
The combination of current collector (often in the form of a grid) and active material contained in a battery.
Depending on the shape and material of the current collector, they result in different battery characteristics. According to the shape of the current collector, the plate is also called a large surface plate, grid plate, tubular plate or box plate.
Several plates of the same polarity are connected together to form a negative (-) or positive (+) plate set.
Plate Pack
The combination of a positive and a negative plate set with the separators in between.
When electrolyte is added, this results in one cell in a battery.

Plate Set
A combination of several plates of the same polarity by means of a pole bridge.
A negative plate set plus a positive plate set plus separators form a plate pack.

Polarity
The arrangement of the two poles of a battery.
Pole Grease
A grease that can protect the poles from corrosion.
For this purpose, it is applied after the battery is connected, around the connection of terminal and pole.
Pole grease, which comes between the terminal and the pole, isolates them from each other and hinders the flow of current!

Positive (+)
One of the two poles of a battery.
The other one is the negative pole. The cap on the positive pole, the positive terminal and the positive cable are usually red. In order for current to flow, both the negative and the positive terminal must be connected to a load.
Positive (+) Plate
A positively charged plate in a battery.
In lead-acid batteries, large-surface, grid or armored plates are often used for this purpose, whose surfaces are designed to carry as much lead dioxide (chemical formula PbO2) as possible.
In a rechargeable battery, the positive (+) plate is called the anode (strictly speaking, the designations anode and cathode depend on the chemical processes and thus the direction of current flow during charging and discharging, but this could lead to misunderstandings, so the designation is determined by the way the current flows during discharging).

Primary Battery
A battery that cannot be recharged.
Pulse Charge
A charging method in which charging takes place with pulsating constant current.
The method is used in the float charge phase. In the currentless charging pauses, the charging voltage can be precisely determined and used as a switching criterion for further charging phases.
Pure Lead Battery
A variant of the AGM battery in which the plates are made of almost pure lead (99.99 %).
This reduces the internal resistance and leads to an extremely high cold start current, is more stable at low temperatures and has a lower self-discharge. In addition, such a battery can tolerate too high charging currents and deep discharge better than other lead-acid batteries.

Rapid-Charge Battery
A battery that can withstand high charging currents.
Recycling Effiency
The degree to which a battery can be recycled.
Lead-acid batteries can be recycled to a high degree.
Up to 95 % of all components of our batteries on this basis can be returned to the production cycle via our recycling partner OneCallCollection.
Resistor
A device with electrical resistance used in an electrical circuit for current control and efficient operation.
Reversed Polarity
The unintended change or reversal of the normal polarity of a battery.
The problem occurs when battery cables or charger cables are connected incorrectly. Chargers with reverse polarity protection prevent the battery from being damaged.
SLI Battery
The battery that starts a vehicle.
During the starting process, a lot of energy has to be supplied quickly. For this purpose, the starter battery supplies power to the starter motor and maintains the supply to all safety-relevant consumers and control elements until the engine starts. Depending on how many consumers are installed in a vehicle, batteries with particularly high cold start currents are used, e.g. AGM batteries. Cars with a start-stop function require batteries that are particularly resistant to cycling. Specially designed AGM or EFB batteries are used here.
In some cars, a backup battery is also installed.

Safety Reserve
The ability of a battery to start even when it is low on charge.
Seal
A kind of sticker that safely closes dry-charged batteries before filling them.
It protects the unfilled battery from unwanted particles and makes it storable for a long time.

Sealed Maintenance-Free
A class of batteries that are firmly sealed, equipped with valves and cannot be opened.
These batteries are leak-proof and can be installed at an angle. They are designed in a way that they lose virtually no liquid and therefore do not have to be refilled.

Secondary Battery
A rechargeable battery.
The chemical reactions that take place when generating electricity can be almost completely reversed when the battery is recharged. There are big differences in quality, depending on how fast you can charge the battery, how efficiently and with little loss it can be charged (i.e. whether you need more power to charge it than you can take out afterwards) and how often you can recharge it before the charge is no longer sufficient to provide the necessary voltage.

Self-Discharge
The reduction of the voltage of a battery when it is not in use.
Charging or discharging always takes place in a battery, and the underlying chemical processes cannot be switched off completely. This means that a battery constantly loses voltage.
The higher the ambient temperature, the higher the self-discharge. In addition, a battery can also lose charge through connected silent loads while a vehicle is not in use.
To compensate for self-discharge, trickle charging can be used, e.g., for batteries that are to remain in continuous operation.
Self-Discharge Rate
The rate at which a fully charged battery discharges without being used.
Rechargable batteries have a higher self-discharge rate than primary cells.
The lower the rate, the longer a battery can be stored without having to recharge it. It also requires only a lower trickle charge to keep the battery fully operational at all times. This is especially important in safety critical applications and for supply batteries.
Separator
An insulator between the positive and negative plates in a battery that prevents the two plates from touching, thus preventing a short circuit.
In addition, the separator can be used to protect the plates from vibrations, making a battery particularly resistant to vibration. For this purpose, separator pockets are used in super heavy-duty batteries, which completely enclose the grid plates. In AGM batteries, the separator made of glass fiber fleece absorbs the electrolyte, which improves the cycle stability and reduces the internal resistance, and particularly high cold start performance is achieved.
Series Circuit
An arrangement in which several batteries of equal capacity are connected together so that the negative terminal of one battery is connected to the positive terminal of the other.
In this circuit, the voltages of the individual batteries are added together, and the capacity is determined by the weakest link in the series.
The opposite is the parallel circuit.

Series/Parallel Circuit
An arrangement of batteries in which some cells are connected in series to increase the total voltage and some are connected in parallel to increase the total capacity.
Service Life
The time between the delivery of a battery and its end of life (EoL).
A battery can still be used beyond this point, but no longer achieves the desired values and must be recharged significantly more often, for example.
Normally, the end of life is measured by falling below a predefined capacity.
The life of a battery depends on many factors, so specifics are usually approximate and only comparable when exactly the same conditions are used. Factors include battery type, battery state of charge, ambient temperature, number of cycles, depth of discharge, and to some extent time, regardless of battery usage.
Shock-Resistant
A feature of batteries that indicates whether they can withstand vibrations particularly well.
Swollen glass fiber separators in AGM-based batteries or the gel-like immobilized electrolyte in GEL batteries ensure that the grids are securely placed and the active material does not loosen from the grid. Thus, it is available for the electrochemical reaction in the battery and the performance of the battery does not drop prematurely. In extremely shake-proof batteries, the plate packs are also crompessed and glued into the case.
There are different standards for vibration resistance: V1 - for standard requirements in passenger car batteries, V2 - for standard requirements in commercial vehicles, V3 - for increased requirements, V4 - for extreme heavy-duty vehicles in factory and commercial use.

Short Circuit
The undesirable direct connection of the two terminals of a battery.
A short circuit caused by a person is life-threatening. Apart from that, short circuits also damage the battery, as they can completely discharge a cell and render it useless.
Short circuits can be avoided by insulation and careful handling. When removing or disconnecting a battery, be sure to remove the negative (-) terminal before the positive (+) terminal and reconnect in the reverse order, tightening the positive (+) terminal first and the negative (-) terminal afterwards.
Silent Loads
Electrical components or devices that draw power from the battery when the vehicle is not running, e.g. computer memory or alarm systems.
Silver
An element used in the positive plates in a silver-zinc battery.
The addition of silver allows for a higher charge voltage.
Smart Charger
A charger that monitors the state of charge of a lead-acid battery and adjusts the power supply accordingly.
Intelligent chargers can also remain permanently attached to the battery without the risk of overcharging or discharging it.
lithium batteries require special chargers.

Spacer
Adapter adhesive pads for installing batteries in larger battery compartments.
Spacers are used, for example, with lithium batteries, which are for the same capacity, significantly smaller than batteries with other technologies.

Specific Gravity
The specific gravity of a substance.
In lead-acid batteries, the density of the electrolyte changes with the state of charge, so in flooded batteries, an acid lifter can be used to determine if it is charged. In a fully charged battery, the acid has a density of 1.28 g/dm3.
Standby
Type of use in which a battery is constantly charged and kept ready for use in an emergency or as a backup.
For this purpose, the battery must be constantly supplied with a trickle charge to counteract self-discharge.
This is important, for example, when used in safety-relevant systems such as uninterruptible power supply (UPS).
Start-Light-Ignition
English term for starter batteries based on the tasks they perform - Start, Light, Ignition.
State of Charge
The degree to which a battery is charged.
A full battery therefore has a state of charge of 100 %.
Stationary Battery
The use of batteries at a fixed location is called stationary.
This includes, for example, batteries that are used as intermediate storage for solar or wind power systems or that supply electricity to smaller stand-alone systems such as in a hut. A further application is as a standby or backup battery in safety-relevant systems such as UPS systems.

Sulfating
An undesirable aging process in batteries in which too much lead sulphate is formed on the electrodes (coarse crystalline sulphation), which cannot be removed even after prolonged charging.
Sulfation reduces the performance of the battery and leads to its failure. Long service life, over-discharge, insufficient charging and acid stratification are causes of excessive sulphation.
Ideally, the chemical process runs as follows and can be completely reversed. In an uncharged battery, the combination of sulphuric acid and lead produces lead sulphate which is deposited as a white layer on the plates. When charging, the hydrogen that is produced removes the lead sulphate from the positively charged plates, and the oxygen combines with the lead to form lead dioxide. When the battery is discharged, lead sulphate is formed again on the plates.

Super-Heavy Duty Battery
A starter battery that is particularly suitable for use in off-road vehicles agricultural, or construction machinery.
In order to withstand the increased mechanical stresses caused by strong inherent vibrations or uneven terrain, batteries for such vehicles are designed to be particularly resistant to vibration, at least in accordance with class V3 (according to EN 50342-1). In order to achieve this, the grid plates are embedded in a special separator pocket made of glass fibre mats and compressed into the housing so that they do not become loose even under extremely strong permanent load and as little active material as possible is lost.

Supply Battery
A battery that enables a self-sufficient power supply, i.e. it stores energy for later, grid-independent, permanent use.
Typical application is the supply of electrical consumers on boats, in mobile homes or buses, where they are installed as an additional battery in order not to overburden the starter battery. In standby applications they take over the power supply if the main power source fails. And they serve as a buffer storage for solar and wind power plants.
A battery for these applications must be particularly cycle resistant.
Apart from this, there are supply batteries in almost all battery classes. As these are mostly stationary applications, wet batteries are also used, especially in the hobby sector and for smaller solar applications.

Temperature Cut-off
A function of battery management systems that regulates the temperature in modern batteries to prevent damage and increase their service life.
Terminal
The electrical connection of the battery to the vehicle circuit.
A battery has one positive (+) and one negative (-) pole. If both poles are connected to an electronic load, electric current flows between them. The location and type of poles should suit the vehicle. If this is not possible, pole adapters are available to attach the cables securely. Only use recommended accessories. All our batteries are marked with the pole type. Pole grease can protect the connection poles from corrosion. There are different types available: Round pole, flat pole, threaded pole, etc., which are noted for each battery.

Terminal Bridge
A component in the battery that connects several plates of the same polarity to form a set of plates.

Terminal Pole Type
The design of the connecting poles of a battery, suitable for the requirements of the respective vehicle or application.

Terminal Voltage
The voltage that can be measured between the two terminals of a battery.
Each battery has an internal resistance. The terminal voltage is the voltage that is actually available after deducting this resistance. For a battery with a nominal voltage of 12 V this should be 12.72 V. If you measure the voltage at the terminal poles and if this value deviates significantly from this value, conclusions can be drawn about the condition of a battery.
Traction Battery
A rechargeable battery designed to power machines and vehicles.
Traction batteries are designed to cope well with many charging cycles, even up to the final discharge voltage, because they are usually charged once and then used until they are empty or nearly empty, and only then recharged. They deliver even amounts of current over a long period of time.
The same properties are needed for stationary applications. They are therefore also well suited as intermediate storage for photovoltaic or wind power systems or for safety-relevant applications that are to be independent of the power grid (uninterruptible power supply/USV).

Tubular Plate
A design in which the positive (+) plates are assembled from several tubes in which the active material is quasi "armored".
Thanks to their design they are extremely resilient and cycle resistant and have plenty of space for as much active material as possible (lead dioxide = PbO2).
The more active material there is, the greater the amount of current a battery can supply. This is crucial for the power capability of a battery.

Uninterrupted Power Supply
A battery operated system that provides standby power when the main power supply is interrupted.
Unwanted Resistance
Resistance in a battery caused by aging processes, e.g. corrosion, or inaccurate manufacturing, which leads to undesirable voltage loss.
Valve-Regulated Lead-Acid Battery
A lead-acid battery that is sealed except for a one-way valve.
The one-way valve releases gas when the internal gas pressure in the battery exceeds the atmospheric pressure by a preselected value.
This makes it possible to produce batteries that do not leak and do not require maintenance. They can also be installed at an angle.
For VRLA batteries, the electrolyte is fixed either in gel or in fleece (AGM). Due to gas recombination, these batteries only emit a small amount of gas and therefore hardly any liquid is lost.

Vent Cap

Vented Battery
A battery where the gases produced can escape.
The gas escapes via one or more vents and is conducted from the engine compartment to the outside via a ventilation hose.
Valve-controlled batteries use gas recombination. In contrast to vented batteries, they are also suitable for inclined.
Venting Hose
A hose that attaches to the vent of a vented battery and drains the gases from the engine compartment.

Vibration
The severe shocks to which batteries are exposed when used in large motorcycles, construction and agricultural machinery.
SHD and HVT batteries are specially designed for such applications.
Volt
The unit of measurement for the electromotive force or voltage of a battery.
Voltage
The electromotive force acting between the terminals of a battery in a closed circuit.
When talking about voltage in batteries, the nominal voltage is usually meant. When determining the state of charge, the open-circuit voltage is measured.
Voltmeter
A device for measuring the voltage in a circuit or for measuring the open circuit or quiescent voltage of a battery.
The open-circuit voltage provides information about the state of charge of a battery.

Watt
The unit of measurement for electrical power. W = Ampere x Volt
Watt-Hour
The unit of measurement for electrical energy. Watt-hour = watt x hours.
Wet Charged
Designation for a battery that is already filled with acid and charged at the factory.
In contrast, dry-charged batteries are only filled with acid and thus activated directly before installation by the retailer or workshop.
Zinc-Air Cell
A dry cell battery system using oxygen and catalyzed carbon as negative plate and zinc as positive plate.
This battery class is often used for electric fences or temporary traffic protection, e.g. by flashing traffic lights.
key-off drain
see → Silent consumers
















































